Investigational Drug or Placebo – Blinding with Transparency
SCHREINER MEDIPHARM
Investigational Drug or Placebo –
Blinding with Transparency
To ensure that clinical trial participants don’t know whether they are receiving the investigational drug or merely a placebo, the substances must be indistinguishable from each other. Schreiner MediPharm has developed a reliable blinding solution for vials used for clinical dosing: due to the use of colored film labels, the different liquids in the vials cannot be distinguished from each other—while the fill level can still be seen.
Before a new active pharmaceutical ingredient (API) can be approved for the market, extensive clinical trials are necessary to test its efficacy and safety. They involve comparative studies in which one group of the participants is treated with the new investigational drug, called verum, while a control group receives a placebo. It is crucial that the trial participants do not know which group is being treated with the investigational drug and which group with the placebo to ensure valid testing of the medication’s efficacy.
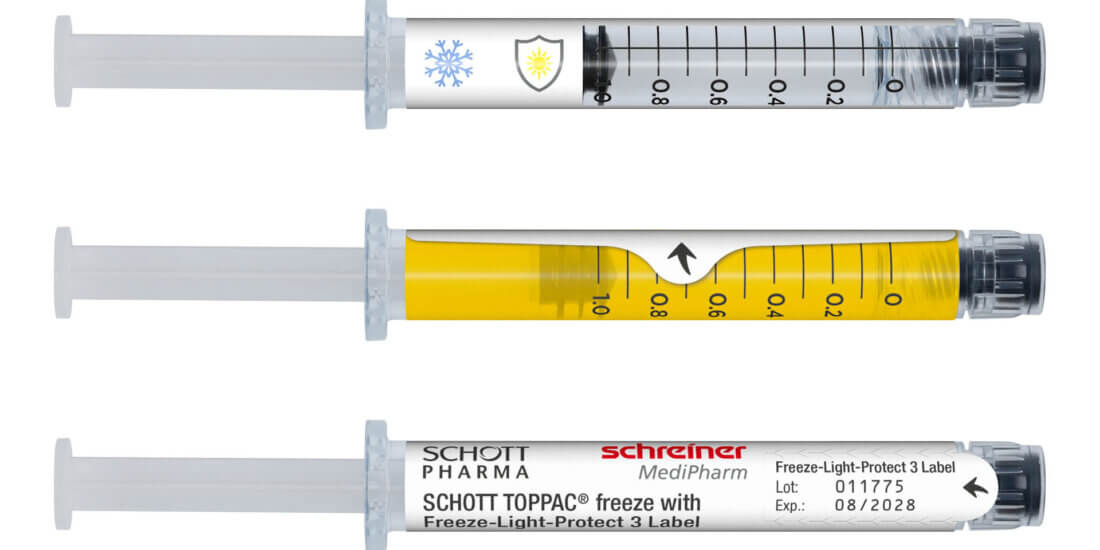
To ensure no one can tell which is the investigational drug and which is the placebo the two substances must not be visually distinguishable from each other. In the case of liquid APIs in transparent vials that is achieved by colored film labels from Schreiner MediPharm. The pharma expert developed them with WuXi STA. Among other things, the company offers clinical supply services for clinical trials and has been producing investigational drugs for clinical trials since 2008. To reliably blind, i.e., neutralize, its liquid investigational drugs, WuXi STA uses the film solution from Schreiner MediPharm.
The blinding solution consists of a self-adhesive label with semi-transparent yellow coloration that was developed precisely for the vials from WuXi STA. The label is transparent enough to see the fill level of the liquid in the vial. The solution from Schreiner MediPharm thus ensures efficient and flexible blinding of the investigational drugs and helps optimize the processes involved in a clinical trial—plus it complies with the requirements of clinical trial regulations.
WuXi STA Clinical Supply Services
WuXi STA Clinical Supply Services (CSS) can provide rapid, agile and reliable supply support for clinical phase I to IV trials and has been manufacturing clinical investigational drugs since 2008. CSS is embedded into the innovative integrated CMC platform of WuXi STA, can truly realize seamless connection from drug product to clinical supply. The customized solution will fully enable pharmaceutical companies to comparator sourcing, primary packaging, secondary packaging, warehouse storage and cold chain distribution, return and destruction to accelerate the process of clinical supply.